Cellular autophagy
The 2016 Nobel Prize for Physiology and Medicine was awarded to the Professor Yoshinori Ohsumi for the discovery of the mechanisms behind cellular autophagy.
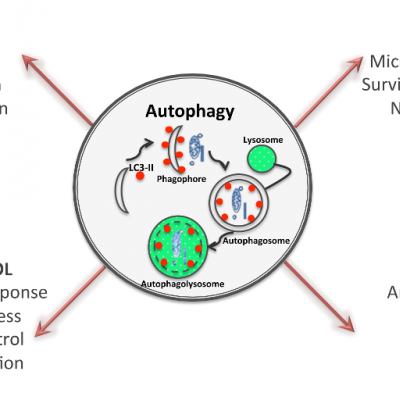
Cellular autophagy is the natural, regulated, destructive mechanism of the cell that disassembles unnecessary or dysfunctional components. It allows for several cellular rôles:
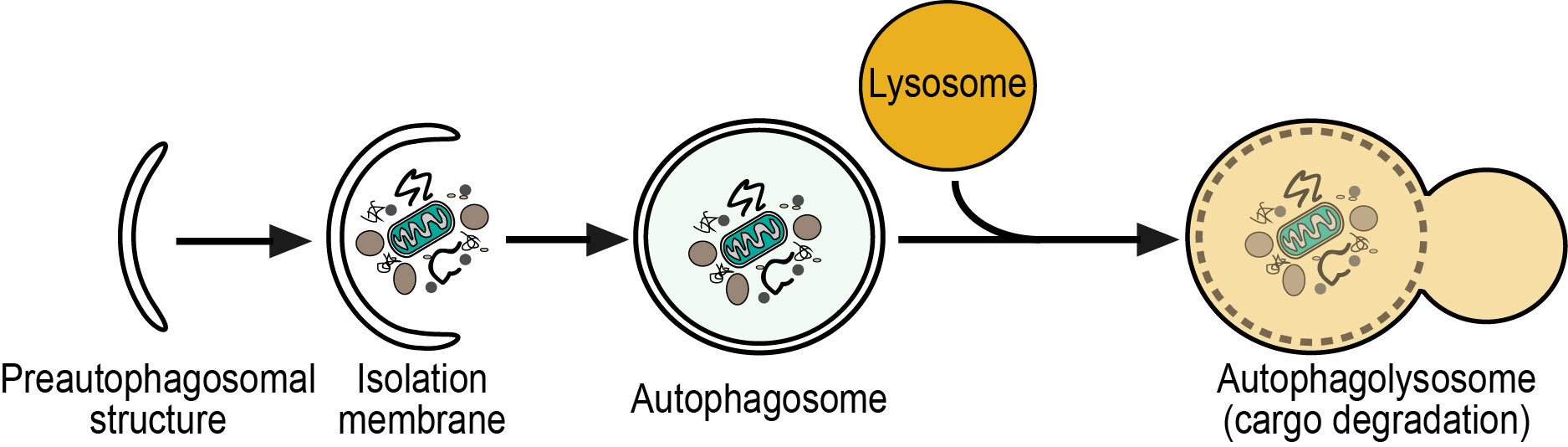
- Cell maintenance - Cellular autophagy (macroautophagy) is critical for cells to survive and to stay healthy. This crucial process ensures the treatment and reuse of waste inside every living cell. In macroautophagy, targeted cytoplasmic constituents are isolated from the rest of the cell within a double-membraned vesicle known as an autophagosome. The autophagosome eventually fuses with lysosomes and the contents are degraded and recycled.
- Coping with starvation - Autophagy has a vital role if an animal is starved, during a seasonal shortage of food for example. During starvation, cells break down proteins and nonessential components or damaged structures and reuse them for energy. In adult humans this process can provide sufficient energy for survival for around four weeks without food.
- Response to infection - Autophagy also plays an essential role in cellular immunity, as it can entrapped, degrade and recycle intracellular pathogens, what can lead to the activation of innate and adaptive immune responses against microorganisms. A variety of host mechanisms exist for recognizing and targeting intracellular bacteria to autophagosomes. However, co-evolution between host-cells and pathogens has selected numerous infectious invading microorganisms able to avoid or usurp autophagy to their own benefit. A subset of viruses and bacteria subvert the autophagic pathway to promote their own replication.
- Metabolism - The cells destroy their own content by enclosing it in membranes, forming vesicles that are then transported to a recycling compartment in the cells for degradation. The disruption of this mechanism is thought to be linked to ageing and to all kinds of diseases such as cancer, infectious diseases, immunological diseases and neurodegenerative disorders. Mutated mice allowed for the study of autophagy and its abnormalities in mammals.
In was in the 1960s that researchers first observed that cells could destroy their own contents in rats and mice, for which they received the Nobel Prize in 1974. But at the time, difficulties in studying the phenomenon meant that little was known about how autophagy worked, what genes were involved, or its role in disease and normal development. That is until Dr. Ohsumi began studying the process in baker’s yeast. It wasn’t until 30 years later that Ohsumi was able to create experiments that identified the genetic and molecular mechanisms essential to autophagy. Using baker’s yeast as a model, Ohsumi showed the underlying mechanisms for autophagy and explained how a similar process occurred in human cells. His work has led to a new paradigm in our understanding of how a cell recycles its content.
From yeast to mammals
It turns out that that in humans and other mammals there are counterparts to almost all the yeast autophagy genes, though the situation is made a lot more complicated by the fact that mammals have more than one copy for each of the genes. Professor Noboru Mizushima of the University of Tokyo made an important advance when, working with Yoshinori Ohsumi, he developed a transgenic mouse in which a protein called LC3 that is found in the autophagosome membrane is fused to Green Fluorescent Protein (GFP – see Nobel Prize for Chemistry 2008) which allowed him and his colleagues to observe and monitor the process of autophage in vivo in mice for the first time.
These discoveries inspired hundreds of researchers around the world to study the process and led to a whole new field and area of inquiry. They opened the path to understanding the fundamental importance of autophagy in many physiological processes, such as in the adaptation to starvation or response to infection.
References:
https://www.nobelprize.org/nobel_prizes/medicine/laureates/2016/
https://www.nobelprize.org/nobel_prizes/medicine/laureates/2016/press.html
http://micro.med.harvard.edu/Micro214/ChoyAutophagyReview_optional.pdf
https://www.nytimes.com/2016/10/04/science/yoshinori-ohsumi-nobel-prize-medicine.html
https://speakingofresearch.com/2016/10/03/nobel-prize-2016/
Last edited: 1 February 2018 16:24