Research in the news
This section is no longer being updated. Last update: Covid-19 in June 2020
-

Alternative methods and animal welfare
27th September 2017
But today, scientists have developed and validated alternative methods shown to lead to safer and more effective products and dr…
-

Animal behavior
27th September 2017
The new animal behavioral studies, from fish to snake, birds and dogs
-
Animal evolution
27th September 2017
Research using fossils, DNA, and other tools to reveal evolution.
-

Asthma
27th September 2017
Asthma is an allergic reaction in the airways of the lungs, and is one of the most common chronic illnesses affecting all age gr…
-
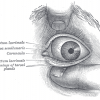
Blindness
27th September 2017
Blindness has several causes but overall results in the incapacity to see. For more information: http://www.animalresearch.info/…
-

The brain
27th September 2017
The brain is an organ that serves as the center of the nervous system in all vertebrate and most invertebrate animals. This part…
-

Cancer
27th September 2017
Cancer is a complex disease and the type of treatments depend strongly on the origin of the cancer and its stage of progression.…
-

Cell, organ and tissue transplants
27th September 2017
Moving cells around within or between different organisms so that they take another function as an individual cell, a tissue, or…
-

Covid-19
15th June 2020
-

CRISPR/Cas9 and gene editing
27th September 2017
-
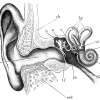
Deafness/hearing loss
27th September 2017
360 million people worldwide have disabling hearing loss. This can be brought on by a variety of causes and most cases cannot be…
-

Diabetes
27th September 2017
Nearly five per cent of the population worldwide suffer from diabetes, with approximately 3.4 million dying as a consequence per…
-

Ebola
27th September 2017
Ebola, is a disease of humans and other primates caused by ebolaviruses. Signs and symptoms typically start between two days and…
-

Heart disorders
27th September 2017
This describes many different conditions affecting the heart. Approximately 600,000 people die from heart disease in the United …
-
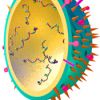
Influenza
27th September 2017
Influenza is a common viral respiratory disease, which is easily passed from between people via small droplets when an infected …
-

Malaria
27th September 2017
Malaria is caused by a single-celled parasite, Plasmodium, which is responsible for at least one million deaths worldwide each y…
-

Microbiota and parasites
27th September 2017
Traditionally, parasite referred primarily to organisms visible to the naked eye, or macroparasites such as worms but the term n…
-

Muscular dystrophy
27th September 2017
Muscular dystrophy refers to a group of genetic, hereditary muscle diseases that cause progressive muscle weakness. Muscular dys…
-

Obesity
27th September 2017
In 2013, the American Medical Association classified obesity as a disease in which excess body fat has accumulated to the extent…
-
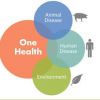
One Health
13th September 2018
-

Pain
27th September 2017
Pain is an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms …
-
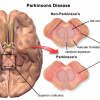
Parkinson's Disease
27th September 2017
Parkinson’s disease is known to be caused by the death of brain cells that produce a neurotransmitter known as dopamine which af…
-

Spinal cord injury
27th September 2017
Every year, about 2000 people in the UK, and several million people worldwide, suffer traumatic spinal cord injury leading to pe…
-

Stem cell therapy
5th September 2018
-

Tuberculosis
27th September 2017
Tuberculosis is a highly infectious disease that we have little power to stop. It is therefore not surprising that 1 in 3 people…
-

Zika
27th September 2017
Last edited: 23 November 2020 11:46

