A scientist at work: exploring the neurobiology of sleep
A scientist at work
Exploring the neurobiology of sleep
Originally published in The American Biology Teacher
Solutions to scientific questions can come from strange sources: the Philadelphian's habit of jaywalking would rank as one. My jaywalking experiences coupled with the memory of one of my long-ago, college swimming races, resulted in the explanation I will give for the organization of REM. But, reading a book on bird behavior really began the process. First, however, a brief review of the characteristics of sleep in general, focusing on that of mammals.
Characteristics of sleep
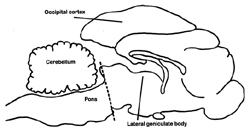
All mammals follow the same scheme with some very rare exceptions. At fairly regular times of the day they adopt a species-specific posture with their eyes closed and become less responsive to environmental stimuliANCHOR. Although humans generally have one major period of sleep, other mammals have more. Recordings from the brain, the electroencephalogram (EEG), reveal a change in the pattern of neuronal activities from that of wakefulness. When an animal falls asleep, the EEG waves change from a low amplitude, high frequency pattern (Fig. 1A) to one with higher amplitudes and lower frequencies (Fig.1B). The waves become even higher and slower as the animal approaches the other major phase of sleep, REM (Fig. 1C). At this transition to REM one can record, with deeply placed electrodes, large waves in a number of structures, very notably in the area receiving information directly from the retina, the lateral geniculate body, and also from areas of the brain that are concerned with alerting. These waves are termed PGO waves. This acronym refers to the three most common sites for recording: the pons, lateral geniculate body and occipital, or visual receiving area of the cerebral cortex (Fig. 2).
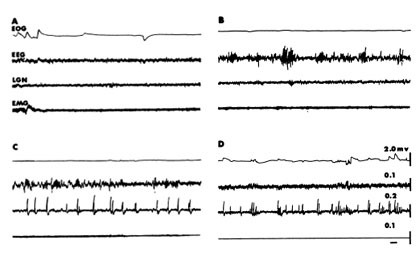
As the animal enters REM two other striking things happen: the EEG changes to a pattern essentially identical to that seen during wakefulness; and skeletal muscle activity, measured as electromyograms (EMG), falls to zero (Fig. 1D). The lack of muscle activity occurs because of an inhibiton of the neurons in the spinal cord that innervate skeletal muscles. However, brief excitatory volleys periodically impinge on the inhibited neurons, resulting in muscle twitches and rapid eye movements. One observes this change in sleep phases about every 25 minutes in cats and dogs and at 90 minute intervals in people. REM comprises about 25% of the total time asleepANCHOR.
A great upsurge in sleep research began with the ground-breaking observations by Nathaniel Kleitman and his students at the University of Chicago in the 1950’sANCHOR. Ironically, their recognition of REM as a “new” state of sleep came at the end of Kleitman's career in sleep research, previously an obscure field. The effect of their discovery was tremendous: within ten years a mob of scientists was puzzling over REM; and within twenty years their efforts led to the development of a new medical specialty, sleep disorders medicine.
Why did REM create such a flurry of activity? Quite simply, it was thought to provide a gateway to the study of dreams; for REM possesses two striking characteristics that excited the imaginations of researchers. The brainwaves recorded by means of electrodes placed on the scalp in people resembled those obtained when a person is awake. Also, the accompanying rapid darting movements of the eyes, which gave REM its name (rapid eye movement), suggested that the sleeper was watching dream events. These two features, once discovered, allowed the researcher to detect REM easily; for the other phase of sleep, prosaically called non-REM, exhibits a brain-wave pattern quite different from REM and wakefulness, as we have noted, and no rapid eye movements. Thus, whenever the REM pattern occurred the researcher could wake up the sleeper and ask if he or she were dreaming and, if so, what was remembered. However, later work has shown that mental activity of varying complexity is also reported when a subject is awkaened out of non-REMANCHOR but the fact remains that REM is an easily identified state of vivid dreaming. Just as more importantly though, its “strange” features induced many more scientists to take an interest in sleep than would have ordinarily.
REM Atonia
I began my own research in 1964 just ten years after the discovery of REM. At that time the skeletal muscle paralysis of REM excited much interest, for it had only been discovered a few years earlier in cats by some French researchers. Indeed, their finding is an excellent example of how science progresses in unexpected waysANCHOR.
They were actually studying the startle reflex in cats, a basic motor response to a noise experienced by everyone. In order to remove the influence of higher brain activity, "thoughts" if you will, they had separated the lower brain leading into the spinal cord from the higher brain (Fig. 2) and were then recording muscle contractions in the cat's neck muscles after a loud noise. However, in between experiments they continued to record the electrical activity of the muscles, which increases or decreases as muscles contract or relax. It was fortunate they did; for a curious thing occurred: Periodically the electrical activity fell to zero. I suspect that at first they may have thought their equipment was not working, that they were observing an artifact, always a concern to a researcher. But they observantly noted that the toes and whiskers twitched during this period just as they do during REM and reasoned that the lower part of the brain sufficed for the peripheral signs of REM to occur.
 Thus, by chance, Michel Jouvet, one of our leading sleep researchers, happened upon his first important discovery because follow-up recordings in normal cats revealed during normal REM the same absence of muscle activity, which we usually refer to as muscle atonia rather than paralysis. Obviously, due to evolutionary pressure, the process of muscle suppression does not occur in the heart muscle; nor does it involve (or only slightly) the diaphragm, or else we would die.
Thus, by chance, Michel Jouvet, one of our leading sleep researchers, happened upon his first important discovery because follow-up recordings in normal cats revealed during normal REM the same absence of muscle activity, which we usually refer to as muscle atonia rather than paralysis. Obviously, due to evolutionary pressure, the process of muscle suppression does not occur in the heart muscle; nor does it involve (or only slightly) the diaphragm, or else we would die.
Yet, atonia of the skeletal muscles during REM is really quite a sensible arrangement. Think of some of your dreams and imagine what kinds of trouble you would find yourself in if you were to act them out. In fact, there are those who do, the majority being older men. In 1983, Mark Mahowald, a neurologist, and Carlos Schenck, a psychiatrist, reported a series of cases in which the patients reported violent activity while they were dreamingANCHOR. Further tests in the hospital revealed that the movements could be correlated with the "actions" in their dreams. Remarkably, about fifty per cent of a more extensive series of patients were normal while awake. Fortunately (for patient and bed partner), these doctors found a medicine that suppresses the violent behavior during REM.
I found a dream report of one of their patients particularly striking, one that well illustrates the "reality" of a dream and the great value of being unable to participate physically. He was sixty-two with memories of past glory apparently, for he had dreamt that he had been playing football. As a running back, he had received the ball for a plunge through the line. But he woke up as he crashed into a defensive lineman while struggling to gain some extra yards. Regrettably, the lineman proved to be his bedroom dresser; and, even more regrettably, he was an old man in his pajamas with the only spectator his horrified wife. (I usually tell this story to counter the flagging interest of high school boys when I speak in schools. They love it, but as a middle-aged man I am the only one in the room old enough to taste its bitter sweetness.)
Mahowald and Schenck recognized this disorder, REM Behavior Disorder, for what it is: a disturbance of REM rather than epilepsy. They did so because of earlier experiments performed in catsANCHOR. Creating a bit of damage (a lesion) experimentally in the lower brain in a region called the pons eliminates the usual muscle atonia, allowing the cats to exhibit various types of behavior in a state we call REM without atonia. This can range from simple head lifting to walking practically normally. The amount of activity released varies from cat to cat and, therefore, from lesion to lesionANCHOR. The degree of complexity of behavior depends on where and how much damage has occurred. Always, though, the cat appears alert to the untutored eye. It will seem to search for something in front of it, orient its head in the direction of a nonexistent sound, pounce on imaginary prey, jump as if startled--all without awakening.
How do we know the animal is not awake? We can wake it up. That sounds like a silly answer, but really says a lot. The cat is in a state from which it can easily be aroused, as in normal sleep and not during an epileptic fit. We perceive the change to wakefulness because the third eyelids, which partially cover the eyes as they normally do in sleep, instantly retract. The animal will then blink if facing a bright light. Moreover, if it is standing on all fours in REM without atonia, the cat will stand more erect when awakened and move gracefully, not awkwardly.
A Collection of Insights
With this background, let me recount how the seemingly unconnected activities I mentioned earlier--reading a book on bird behavior, jaywalking and swimming a 100-yard race--contributed to my hypothesis of REM organization. The first insight came after reading The Herring Gull's World by Niko TinbergenANCHOR, one of the world's leading ethologists. By means of careful, detailed observations of the birds' interactions with each other and other creatures and objects in their surroundings, he developed such insights that he seemed to be able to crawl into the gull's mind. Much that he found depended on paying attention to the gull's various postures and movements. With such understanding, he told us why the birds behaved as they did.
At that time we were studying the role one portion of the brain, the cerebellum, might play in the control of posture, movement and muscle tone during sleep because the cerebellum had long been known to be a regulator of all three during wakefulness. I found little to interest me when the cats were sleeping as I sat in front of our recording machine watching the pens trace out rather unimaginative (apparently unchanged from normal in other words) lines on the paper flowing beneath them. That may have been the end of it had I not happened to be reading The Herring Gull's World. I thought to myself: What if I imitate Tinbergen and watch my cats while they are sleeping rather than watching the pens? Would their behavior reveal something our recording machinery did not?
Now watching a cat sleep may seem as exciting as watching corn grow--and it usually is--but this time was different. Just before the cat entered REM, a series of jerks of the legs occurred, which stopped as the atonia of REM ensued. Not much, one would say; but inspired by Tinbergen I was able to make something of these seemingly inconsequential movements.
In the first place, they differed from cat to cat: In some the elbow flexed and in others it extended. We reasoned that the cerebellum, which works to prevent movement from being too excessive in one direction or another (preventing a "drunken" staggering back and forth in other words), had been damaged by our surgery in a way that allowed too much movement in one direction or the other even in sleep. In fact, a cat that suddenly flexed its elbow in the period just before REM also did the same when it was awake and walking. Whether there was hyperflexion or hyperextension depended on the location of the cerebellar lesion, for the body’s musculature is represented in orderly way in the cerebellum. So, we reasoned, modulation by the cerebellum normally allows the muscles to settle smoothly into the atonic state of REMANCHOR.
The story could have ended with that rather mundane conclusion. However, the young, observant veterinary student working with me, Robert Bowker, happened to drop his keys on the floor while recording and then noted that the same leg that jerked spontaneously just before REM also responded with the same movement in non-REM when the keys dropped. He also observed that a large PGO-like wave appeared in the tracing (Fig. 3). PGO waves also occurred during REM in response to a noise, while atonia prevented leg movement. He may well have missed the wave initially, though, if there had been no accompanying jerk to signal when the noise occurred.
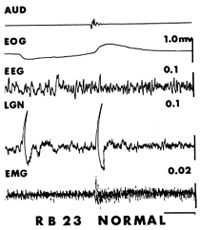 This simple observation led directly to our understanding of a prominent phenomenon of REM that many had puzzled over, the PGO wave, and also set us on the path to understanding much more about REM. Although we then found that sounds also elicited PGO waves before REM in cats without cerebellar damage and during REM as well in between those that occur spontaneously, we were still stuck on the idea that they were somehow linked with movement.
This simple observation led directly to our understanding of a prominent phenomenon of REM that many had puzzled over, the PGO wave, and also set us on the path to understanding much more about REM. Although we then found that sounds also elicited PGO waves before REM in cats without cerebellar damage and during REM as well in between those that occur spontaneously, we were still stuck on the idea that they were somehow linked with movement.
Again, a student was key to advancing the story. I had assigned a particular reading to a Penn undergraduate, Peter Reiner, doing an independent study with me. In the articleANCHOR the author referred to a drug that had created insomnia and had also released PGO-like waves even during wakefulness. He had remarked that each time a wave appeared the cat acted as if it had been startled. Reading that passage with my student led to a sudden insight. I ran down the hall to Bob to tell him what we had read and had concluded: "We're startling our cats with noises. So REM with all its PGO waves must be a period when the brain is startling itself continuously."
Of course, we had to admit that the spontaneous "startling" represented by PGO waves does not awaken cats, but then neither did our noises arouse our cats. Then, I thought of the behavior of cats in REM without atonia. They, too, seemed to startle spontaneously and even orient (direct their attention) to nonexistent objects. And, they did that throughout each REM episode. We then generalized our terminology by adopting the term “alerting” to express our sense of what the PGO wave represents.
By the experimental trick of lifting the body out of paralysis in REM by eliminating the motor inhibitory mechanism, one demonstrates in a striking way one of the fundamental features of REM: "alertness" during sleep. This does not mean that one is more easily aroused from REM than from non-REM, for much evidence points to the contrary. The point is that the behavior observed in REM without atonia, added to the insights gained from recording PGO waves (not to mention the pattern resembling wakefulness on the EEG), suggests that the REM brain most resembles the awake brain when a person is on high alert. (An as-yet-uknown mechanism prevents actual arousal.)
All of this pointed to one obvious conclusion: The paralysis of REM is secondary to the brain's highly "excited" condition because we can eliminate the paralysis and still have REM appear. Although it is quite useful that we are paralyzed in REM -- just remember some of your dreams – REM without atonia demonstrares it is not necessary.
Thinking about how similar these two obviously different states of being, alert wakefulness and REM, are in a number of ways, I was clearly primed to pay attention to what I did when something unexpected happened, when I was surprised or startled -- which brings us to jaywalking.
Commuters to the University of Pennsylvania gather to enter a major gateway to the campus at 34th and Walnut Streets. They must cross Walnut. Typically, pedestrians cross well before the light changes to green because, like them, the traffic tends to move in bunches and must traverse a very long block before reaching 34th Street. Only the occasional, delinquent car or bus will threaten their premature crossing; but clearly the group, at least some in it, will have calculated the distance between it and that oncoming vehicle and judged it still safe to cross.
But what about the straggling jaywalker lagging a few yards behind that band? What if he is a professor lost in thought? What if he takes the cue to cross from their behavior without making his own calculations? Well, he still has time not to go forward, but what happens just before that decision is what interests us. Just as the car enters his peripheral field of vision, he will sense a slight give in the knees or, if already walking, a slight "hitch" in his stride as the leg that is moving forward stops moving for a fraction of a second. I have sensed this on too many occasions to doubt that it happens.
I happened to have one of those straggler's experiences not long after we gained our insight into REM as a period when the brain acts as if it were continually receiving alerting signals. When I safely reached the other side of the street, I again had a sudden flash of insight: The paralysis of REM depends on the brain's "alerting" just as the hesitation in movement does when we are awake -- but in exaggerated form. That is just the way it happened: I'm not making up a convenient story.
Furthermore, an experience from the past flooded my mind. It was a dramatic experience I had had as a college swimmer during a 100-yard freestyle race, which is a flurry of action over very quickly. In a 25-yard pool the swimmers make three turns as efficiently as possible. In this particular race I could see I was in the lead going in to the final turn and, thus, was very excited. But then, a very peculiar thing happened as I completed the last turn: I stopped dead in the water; I could not move a muscle. In the water churning with tumbling bodies I felt paralyzed as if in a bad dream (Actually, awareness of difficulty in moving appears in some dreams, which is quite likely the result of feedback information from the inhibited muscle-control system). It could only have been a second or so, but by then everyone had left me behind. Even then I knew that tightened muscles had not stopped me: I just had no muscle power at all. Something had over-loaded my circuits and short-circuited the energy to them. I had no idea why.
But now I know that I had experienced a brief paralysis brought on by the excitement of actually winning a race, a gross exaggeration of what transpires when one is suddenly surprised (excited) by an unexpected stimulus. So thinking back on that moment, while standing at 34th and Walnut, brought it all together. The atonia of REM is an inevitable consequence of the peculiar high level of excitement within the brain -- peculiar because it actually occurs within sleep. And because REM will occur without the atonia in our experimental animals and people with REM Behavior Disorder, the excitement generated within the brain during REM must not depend on the paralysis to keep an individual from awakening. As we have noted above, what the actual mechanism is is not known.
I suggest that the linkage between excitement and suppression of motor activity that seems to be shared by two grossly different behavioral states is an example of Nature's parsimony and, therefore, efficiency. Why evolve two parallel systems when one (with some modifications depending on the particular demand) will do? After all, the same muscles we employ to move us from one point to another serve to increase our body temperature by shivering when we are cold; and the lungs, chest muscles and diaphragm that participate in respiration also help us eliminate heat during panting.
The nervous system evolved to allow an animal to react in an automatic manner when the brain is very excited. When we are awake, an unexpected stimulus is always potentially life threatening, so Nature has installed a little "look before you leap" program. There is always a brief moment of hesitation, orienting the head toward the stimulus and then a rapid assessment of the situation before appropriate action is taken. This could be "run," "approach for closer inspection" or "dismiss as unimportant." Various factors come in to play during the assessment. For example, a prey animal must decide if the stimulus is a predator and, if so, how far away it is before deciding to flee or ignoreANCHOR. And why not use the same mechanism whenever running about willy-nilly in sleep would be disadvantageous? Patients with REM Behavior Disorder can tell us about the disadvantage of not being paralyzed in REM.
So, there is in the brain an "automatic" connection made between a high level of excitation with a system that puts a "clamp" on action, however brief the latter might be. The peculiar characteristics of another sleep disorder called narcolepsy, which afflicts about .05% of our populationANCHOR, support this hypothesis. These patients are miserably sleepy all the time. In addition, various normally arousing situations or stimuli, e.g., laughter, fright, anger, startle, induce them to collapse as I did during my race -- but tragically for them, many times during the day. The bouts of paralysis, called cataplexy, often continue directly into regular REM episodes with the usual paralysis or atonia continuing, of course. Thus, rather than following the normal sequence of waking to non-REM to REM, the narcoleptic often shifts directly from waking into REM. Instability must exist that allows an abnormal switching between the two different modes of the excitement-motor suppression system. Where that instability is in the brain remains a big unknown.
But step away from the science for a moment and think of suffering an attack while sitting in the bathtub or driving a car. Can you appreciate the danger and inconvenience these folks face in addition to the misery of being sleepy all of the time? As yet, we only have medicines that lighten their load a bit, neither curing them nor alleviating all of their symptoms.
Conclusion
To conclude, ideas can spring from many sources. An observation made during the course of an experiment in one field or, as demonstrated here, during a life experience, may be the key to an advance in another. Individually, the observations by themselves may not be all that exciting, but something about them may stimulate a thought that leads in a new direction – as Tinbergen's observations of gulls' movements triggered an idea that led to a new way of thinking about PGO waves and, ultimately, REM.
Acknowledgements
Research in the author's laboratory was supported by NIH grants MH42903 and NSl3110. I also thank various individuals who have supported my public education efforts.
References
- Hendricks, J.C., & Morrison, A.R. (1981). Normal and abnormal sleep in mammals. Journal of the American Veterinary Medical Association, 178:121-126.
- Jouvet, M. (1967). Neurophysiology of the states of sleep. Physiological Reviews, 47, 117-177.
- Dement, W.C. (1994). History of sleep physiology and medicine. In M.H. Kryger, T. Roth, & W.C. Dement (Eds.), Principles and Practice of Sleep Medicine. (pp. 3-16). Philadelphia: W.B.Saunders.
- Pivik, R.T. (1994). The psychophysiology of dreams. In M.H. Kryger, T. Roth, & W.C. Dement (Eds.), Principles and Practices of Sleep Disorders Medicine. (pp. 384-393). Philadelphia: W.B. Saunders.
- Morrison, A.R. (1993). Animals in sleep research. In M. Carskadon (Ed.), Encyclopedia of Sleep and Dreaming. (pp. 38-39). New York: MacMillan.
- Mahowald, M.W., & Schenck, C.H. (1994). REM sleep behavior disorder. In M.H. Kryger, T. Roth, & W.C. Dement (Eds.), Principles and Practices of Sleep Disorders Medicine. (pp. 574-588). Philadelphia: W.B.Saunders.
- Morrison, A.R. (1996). Contributions of animal models to sleep disorders medicine. Lab Animal, 25:22-26.
- Hendricks, J.C., Morrison, A.R., & Mann, G.L. (1982). Different behaviors during paradoxical sleep without atonia depend on pontine lesion site. Brain Research, 239, 81-105.
- Tinbergen, N. (1971). The Herring Gull's World. New York: Basic Books, Inc.
- Morrison, A.R., & Bowker, R.M. (1975). The biological significance of PGO spikes in the sleeping cat. Acta Neurobiologiae Experimentalis, 35, 821-840.
- Dement, W.C. (1969). The biological role of REM-sleep (circa 1968). In A. Kales (Ed.), Sleep: Physiology and Pathology. (pp. 245-265). Philadelphia. Lippincott Co.
- Ratner, S.C. (1967). Comparative aspects of hypnosis. In J.E. Gordon (Ed.), Handbook of Clinical and Experimental Hypnosis. (pp. 550-587). New York: MacMillan.
- Guilleminault, C. (1994). Narcolepsy syndrome. In M.H. Kryger, T. Roth, & W.C. Dement (Eds.), Principles and Practices of Sleep Disorders Medicine. (pp. 549-561). Philadelphia: W.B.Saunders.
Last edited: 15 October 2014 12:14
