Multiple sclerosis
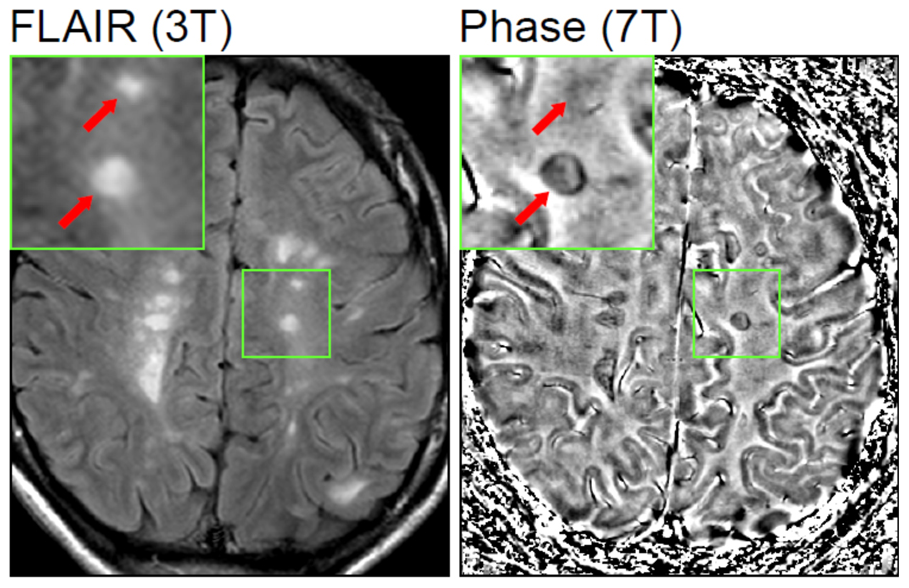
Multiple sclerosis (MS) is a potentially disabling autoimmune disease of the brain and spinal cord (central nervous system). In patients with multiple sclerosis, the immune system attacks the protective sheath (myelin) that covers nerve fibers. The scaring and destruction of the protective layers of neurones, and potentially the underlying nerves, means that messages travelling along the nerves become slowed or disrupted. This causes incorrect transmission of nerve impulses between neurons, and communication problems between the brain and the rest of your body.
Because different nerve cells can be affected from one patient to the next, patients with MS experience a wide range of potential symptoms, including fatigue, muscle weakness, problems with vision, speech, arm or leg movement, sensation or balance. Sufferers usually begin by entering a relapsing-remitting phase of 10-15 years, during which they experience periods of symptoms followed by apparent recovery. However, as the disease progresses into the primary and secondary phase, the individual experiences worsening symptoms with no remission.
MS is one of the most common causes of disability in younger adults in particular women. It is most commonly diagnosed in people in their 20s, 30s and 40s - although it can develop at any age – and is about 2 to 3 times more common in women than men. Research even suggests the proportion of women with MS is increasing. An estimated 2,500,000 people in the world have multiple sclerosis. The distribution of MS around the world is uneven. Generally, the prevalence increases as you travel further north or south from the equator. An estimated 130,000 patients, most of whom are women, live with MS in the UK. Nearly 7,000 people are newly diagnosed each year. This means that around one in every 500 people in the UK has MS, and that each week, 130 people are diagnosed with the disease.
Exactly what causes the immune system to act in this way is unclear, but most experts think a combination of genetic and environmental factors is involved. Although some forms of treatments exist that slow the progression of multiple sclerosis, currently, there is no cure.
Existing treatments
Animal models
Stem cells and transplants
Harnessing the body's own repair mechanisms
Targeting the immune system
Genetic aspects
Drug treatments
Existing treatments
There's currently no cure for multiple sclerosis, but a number of treatments can help control the condition and ease symptoms. A large body of research on the immunobiology of MS, which include studies in animals, has resulted in a variety of anti-inflammatory therapies that are highly effective at reducing brain inflammation and clinical/radiological relapses. However, despite potent suppression of inflammation, benefit in the more important and disabling progressive phase is extremely limited; thus, progressive MS has emerged as the greatest challenge for the MS research and clinical communities.
Choice of treatment depends on specific symptoms and difficulties. It may include:
- treating relapses with short courses of steroid medicine to speed up recovery
- specific treatments for individual MS symptoms
- treatment to reduce the number of relapses using medicines called disease-modifying therapies
Disease-modifying therapies may also help to slow or reduce the overall worsening of disability in people with a type of MS called relapsing remitting MS, and in some people with types called primary and secondary progressive MS, who have relapses. The majority of DMTs approved by the Food and Drug Administration (FDA) since the early 1990s are effective at helping to manage relapsing-remitting MS, which affects between 85% and 90% of people diagnosed with this disease.
Unfortunately, there's currently no treatment that can slow the progress of primary progressive MS, or secondary progressive MS, where there are no relapses.
- Natalizumab
Natalizumab, sold under the brand name Tysabri among others, is a medication used to treat multiple sclerosis and Crohn's disease. It was been licensed in the UK and the US for rapidly evolving, severe, relapsing remitting MS. It is a recombinant humanised monoclonal antibody produced in murine myeloma cells and targeted against the cell adhesion molecule α4-integrin. It inhibits the transmigration of immune cells into the inflamed parenchyma of lymphatic organs and the central nervous system (CNS).
Natalizumab was the first mAb approved for therapeutic trials in MS and now it represents a second-line drug for treating MS.
In 1994 antibodies targeting immune system cells were shown to halt the development of disease in mouse models of MS, specifically mice with experimental autoimmune encephalomyelitis (EAE) 6. The antibodies used were mouse antibodies and couldn’t be transferred and used in humans without causing an immune response. Researchers then went on to producing humanised antibodies that would work in human patients, today known as Natalizumab. It was shown to be effective in preventing EAE.
Once natalizumab had been proved to be effective in this model system in animals, it was tested on humans in several clinical studies in the 1990s. The antibody reduced the frequency of permanent damage to the brain by almost 45 percent, and the frequency of flare-ups declined by almost 70 percent.
Natalizumab came onto the market in the USA in 2004. Following its temporary withdrawal due to a fatal complication, natalizumab was licensed again in the USA in 2006 and, finally also in Germany for the treatment of highly active, relapsing multiple sclerosis.
- interferon betas
Interferons are medicines that "interfere" with diseases that attack the body. They may work by decreasing inflammation and increasing nerve growth. There are many interferon drugs.
Drugs called interferon betas are common treatments for multiple sclerosis (MS), reducing relapses and slowing motor function decline. Interferon beta has been approved for the treatment of relapsing–remitting multiple sclerosis and secondary progressive multiple sclerosis. Interferon beta, a protein known to contain a zinc binding pocket, is thought to reduce proinflammatory molecules and even increase production of anti-inflammatory species in MS patients. Numerous animal studies have led to the understanding of this molecule and the drug today used to treat MS. Studies are still ongoing to understand how it works.
Over the years, an increasing number of interferons have been discovered along with their many different biological activities. Interferons have become part of an interacting family of biological response-modifying proteins. Because of the complexity of these systems, animal experiments are the only way to assess the clinical potential of interferons(and interferon-like molecules).
These molecules have benefitted human, but also animal health. A number of studies have evaluated the use of human, canine and feline Interferons as treatments for infectious, inflammatory and neoplastic disease in dogs and cats.
- monoclonal antibody ofatumumab
The monoclonal antibody ofatumumab (Kesimpta, Arzerra), approved by the FDA in 2020, targets cells that damage the nervous system. These cells are called B cells. Ofatumumab, which is given as an injection under the skin, decreases multiple sclerosis brain lesions and worsening symptoms.
It was extensively vetted in animal models, particularly mouse models. More information here: https://www.ema.europa.eu/en/documents/assessment-report/kesimpta-epar-public-assessment-report_en.pdf
- glatiramer acetate
The glatiramer acetate preparation is a random polymer consisting of repeated sequences of four amino acids, which has been shown to suppress EAE progression in mice, probably through the stimulation of Th2-mediated anti-MBP immune response or by inducing killing of antigen-presenting cells (APCs) and generation of Treg cells.
- Mitoxantrone
Mitoxantrone was first proven to be a powerful immunosuppressive drug in EAE in mice, and it is now a second-line component of MS therapy. Cytotoxic effects on lymphocytes and induction of apoptosis of APC have been proposed as the major mechanism of action of this drug.
For more information on treatments against MS : https://www.mayoclinic.org/diseases-conditions/multiple-sclerosis/expert-answers/emerging-treatments-for-ms/faq-20096786
Animal models of multiple sclerosis
So far MS is thought to be unique to humans, as other species do not appear to develop this spontaneous disease.
Research using animals is vital in advancing our understanding of MS. And it's been critical in the development of new treatments. Many current disease modifying therapies (DMTs) were only possible as a direct result of testing and experiments involving animals. By law, new compounds must be shown to be safe and effective in animal studies before they can be tested in human clinical trials.
Since MS is a complex disease, there is no a single animal model that can capture the entire spectrum of heterogeneity of human MS and its variety in clinical and radiological presentation. They have been an importanttesting tool to study disease development and novel therapeutic approaches.
The three most characterised animal models of MS are
- the experimental autoimmune/allergic encephalomyelitis (EAE);
- the virally-induced chronic demyelinating disease, known as Theiler׳s murine encephalomyelitis virus (TMEV) infection
- the toxin-induced demyelination.
All these models, in a complementary way, have allowed us to reach a good knowledge of the pathogenesis of MS, although they are not perfect, and do not perfectly reflect all the aspects of the human disease. In particular, disease initiation is usually highly artificial in the animal models, and the time-frame of disease progression slightly off.
A great number of animal models, developed for MS, have garnered consistent criticism, often resulting in disappointing failures. It is important to remember that there is no a single animal model that can reflect the entire spectrum of heterogeneity of MS and this research field lacks a focused disease model for progressive MS. To approach the complexity of MS, current progress in humanising the entire immune system in rodents will possiblyprovide substantial advantages for exploring novel immune-modulatory approaches.
Despite the clearly existing limitations, animal model of EAE will continue to play a key role as a first-line model system in the development of novel therapeutic approaches for MS, especially for shedding light on specific mechanistic questions. Basic science on MS will continue to rely on these models for new drug development and for a better comprehension of the different pathogenic mechanisms of MS.
And although it has become clear that rodent and human immune systems have profound differences (as they are evolutionarily distant), they share some essential principles and, in this context, the availability of three major animal models of MS allowed the understanding of relevant features of the human MS. EAE is the model which currently better reflects the autoimmune pathogenesis of MS and is extremely useful for studying experimental treatments.
- Experimental autoimmune encephalomyelitis (EAE)
Experimental autoimmune encephalomyelitis (EAE) is the most commonly used experimental model for the study of MS. Autoimmunity to central nervous system (CNS) components is induced in susceptible mice through immunisation with self-antigens derived from basic myelin protein.
Experimental autoimmune encephalomyelitis (EAE) mice was first described 50 years ago. Rivers et al. (1933) first described, in monkeys immunised with rabbit brain extracts, paralyisis associated to perivascular infiltrates and demyelination in the brain and spinal cord, as acute disseminated encephalomyelitis, later called experimental autoimmune encephalomyelitis (EAE). Experiments were also performed in other animal species such as guinea pigs, monkeys (Kabat et al., 1947, Morgan, 1947); however mice and rats resulted the best model to evaluate acute monophasic, relapsing–remitting and chronic progressive EAE through immunogenetic, histopathological and therapeutic studies.
Mice are the most common species used in EAE research, which can be largely attributed to their low cost, availability of research tools and the ability to generate transgenic strains. C57BL/6 mice are one of the most popular strains used in biomedical research. The SJL mouse is another strain frequently used for EAE induction. Lewis rats are the most popular strain of rat used in EAE research.
Using this model, researchers can produce acute, chronic, and relapsing-remitting EAE models to trial MS treatment. The model also allows the nature of the disease to be studied as it resembles the pathological features of human MS in many aspects including inflammation, demyelination, axonal loss, gliosis and immune reaction. EAE model significantly contributed to our knowledge of autoimmunity and neuroinflammation, changing the course of MS understanding and thus allowing the development of novel therapeutic approaches for this disease.
EAE models have historically been used pre-clinically to assess and define the utility of novel MS therapies. The spectrum of agents showing promising results in EAE is extensive and ranges from natural compounds to modern genetic manipulation of the immune system with cytokines and antigen. The most important examples in this context are represented by glatiramer acetate, mitoxantrone and natalizumab.
Nonetheless, there are also several limitations to the use of this animal model because of the differences in the pathogenesis of EAE compared to that of MS. The model has been criticised because of poor translation in terms of predicting treatment efficacy. The major difference in EAE disease compared to MS is the induction process. In EAE, inflammation and blood–brain barrier (BBB) disruption are induced after external immunisation against myelin antigens boosted with adjuvants from bacterial origin. This could lead to fundamental differences in the priming and activation of inflammatory factors. Furthermore, some inflammatory profiles don’t fit. in EAE models, CD4 T-cell-mediated inflammation is dominant, whereas, in MS, the CD8 component is more important.
More information: https://onlinelibrary.wiley.com/doi/10.1002/jnr.24224
- Theiler׳s murine encephalomyelitis virus (TMEV)
Like other autoimmune diseases, it is thought that MS can be induced by a viral infection. This theory has been supported by animal studies and has notably led to an important animal model of the disease.
Viral infections of the CNS can induce demyelination in mice, just like it is seen in human patients with MS. The best studied are the picornavirus, such as Theiler׳s murine encephalomyelitis virus (TMEV) and certain strains of the coronavirus, such mouse hepatitis virus (MHV).
TMEV represents one of the neurotropic viral infection models for MS. The TMEV model and EAE display several important differences, such as a requirement for viral persistence, immune system activation, neuropathogenetic mechanism, and clinical courses. Unlike EAE, the disease is always chronic-progressive in susceptible mice and TMEV can induce inflammatory demyelinating disease only in mice and not in other species, such as rats and primates.
Contrary to MS, in TMEV infection, axonal damage precedes demyelination (inside-out model) and the distribution of damaged axons observed during the early phase corresponds to regions, where subsequent inflammatory demyelination occurs during the chronic phase. The pathogenesis of TMEV-induced demyelination in part differs from that in human MS, where persistent viral infection of the CNS has not been demonstrated.
Other viruses have also been used to cause MS-like disease in mice. A different mouse model employs the murine herpesvirus (MHV). This virus infects and replicates in oligodendrocytes leading to demyelination both through a direct cytotoxic effect of the virus and through immune-mediated mechanisms. Other viral MS models also include the Semliki Forest Virus (SFV) and Sindbis Virus (SV) models.
- Toxic models of MS
While EAE is the most commonly used model to reflect the autoimmune origin of MS, toxic demyelination is more suitable to study the de- and re-myelination processes. Toxin induced demyelination models, compared to EAE and virus induced demyelinating syndrome, do not reflect MS disease, but are mainly established systems to study the process of de- and remyelination.
Cuprizone and lysolecithin are the most common agents utilised to induce demyelination.
Cuprizone is a copper chelating reagent which, supplemented to normal rodent chow, causes oligodendroglial cell death with subsequent demyelination, together with a profound activation of astrocytes and microglia. Although not appropriate to study autoimmune mediated demyelination, the cuprizone model is a suitable tool to study basic mechanisms during de- and re-myelination in absence of primarily immune-mediated phenomena.
Lysolecithin is an activator of phospholipase A2 which induces focal areas of demyelination upon injection into the spinal cord in several animals, including cat, rabbit, rat and mouse. Lysolecithin exposure has been used to induce demyelination in adult zebrafish (4–7 months of age), as has been performed in murine models.
- a model for late stage multiple sclerosis
Multiple sclerosis usually evolves with age. The first or early stage of MS, that usually occurs at a younger age, is called the relapsing-remitting stage, which means that symptoms come and go. This stage of the disease is quite well understood and treated. It is very much driven by inflammation in the brain.
As patients age, the nature of the disease changes and the immune system becomes less important in the progression of MS. After the age of 40, it is normal for nerves to begin to die in the brain and spinal cord. But if the nerves are already damaged by MS or, more specifically, their protective myelin shield is, they die faster and patients get gradually more disabled over time, in a progressive way.
“For years now, we’ve been trying to find a way to keep these neurons alive,” says Prof Williams. “We’ve been looking at post-mortem brains but this has limitations. They only give researchers a view of a unique time-point. So scientists have often turned to animal models. But most of the models for MS are inflammatory models of early stage MS.”
“The EAE mouse model really isn’t a good model for looking at the neurodegeneration side of the disease. In general, the later stage models are fairly poor representations of the human disease. They don’t answer the right questions. That is the key to modelling any disease. You have to use the right model to answer the right question that you can then apply back to humans. We wanted to reproduce the lack of myelin in the brain. To do so we made a small 2mm hole in the skull of mice and placed a small sponge soaked in a de-myelinating substance at the surface of the brain."
The de-myelinating substance is effectively a detergent. It removes the myelin from all of the neurons in the area. This causes a particular set of neurons to die, just like in MS. These are the inhibitory neurons, the ones that limit and control the actions of the other brain cells. Inhibitory neurons have to fire very fast and often and so need more energy that other neurons, energy that the myelin sheath usually helps to provide.
"This affected only inhibitory nerves of the brains, not the excitatory one, just like in MS. This is effectively the first realistic mouse model of human MS.” According to the researcher.
Although the operation sounds severe, the mice barely develop any symptoms. The operation takes about 20 minutes and only a very small part of the cortex is affected. The lesion is very limited and distinct. The animals recover from the operation very quickly and don’t need special care. They just need to be kept warm while they recover from surgery, but after they can feed and run around and do everything as usual. As such, the procedure is classified as mild to moderate.
Prof Williams and her team are expecting a lot from this model: “We’re hoping it will change the pace in neurodegenerative research, which has been very slow and rather unsuccessful. We’ve been working on trying to keep nerves alive for years, decades, probably half a century, and not found anything. Hopefully this model will change things, for MS patients but also patients with other diseases that cause nerves to die.”
- Fish model of MS
Zebrafish models of MS are emerging as a useful research tool to study MS and validate potential therapeutic candidates due to their rapid development and amenability to genetic manipulation. This makes them excellent model systems for investigating neurological disorders and developing high-throughput drug screens, something not possible in current in vivo models.
In particular, the similarities in myelin and oligodendrocyte biology, alongside the rapid development and large numbers available, give them potential to be used as an excellent model for in vivo screening for pro-myelinating therapeutics. Currently, zebrafish models are at an early stage of development and the primary advantages of zebrafish models are on the characterisation of the mechanisms of demyelination and remyelination. Further work on the immune system involvement in the modulation of demyelination would be required to ascertain the strengths and weaknesses of zebrafish models in MS research. However, it is important to understand that zebrafish immune system does not mimic the mammalian system.
Despite some differences, the zebrafish CNS is a simplified version of the mammalian nervous system and contains the majority of the cell types, connections and structures of the mammalian brain and spinal cord. Zebrafish are emerging as an excellent model for looking at the cell–cell interactions during myelination. Using different genetic mutants and transgenic reporter lines, it is possible to visualise the processes of myelination and demyelination in real time. These zebrafish models have been used to identify some interesting insights into the mechanisms of myelination.
The majority of work undertaken so far using zebrafish as a model has focused on investigating the mechanisms of remyelination and OPC differentiation, using toxin-induced demyelination models and genetic cell ablation models. Unfortunately, this model requires surgery and a relatively long time for recovery making its applicability for drug screening limited, but would be a powerful tool to validate potential therapeutics from primary screens. A genetic inducible demyelination model has also been generated by the ablation of oligodendrocytes and thus removal of myelin.
Indeed, zebrafish can naturally repair defective nerve tissue. Although their regenerative capacity belongs only to them, many genes involved in this process also exist in a similar form in humans. In principle, agents that boost these repair genes in fish could potentially also work in humans. However, the differences between the genetic makeup of fish and humans are often significant. The larvae are therefore sometimes of limited use in the search for new drugs. To overcome this problematic, some researchers have made larvae fish a bit more 'human-like'. This humanisation could make the search for active pharmaceutical substances much more efficient.
More recently an EAE zebrafish model has been developed for developing rapid screens in vivo. Humanised zebrafish larvae allow many substances to be screened quickly, and with a high chance of success, since the target genes originate from humans. It is a very promising avenue for drug development. Once singled out, the substances are first tested in cell cultures. Only individual, very promising candidates are then tested in mice or other animal models.
The ultimate goal in the future is for zebrafish models to be used as a preclinical screening tool to triage compounds in drug screens prior to screening in higher models.
For more information : https://journals.sagepub.com/doi/full/10.1177/1352458518805246
Non-human primate models
The pathogenic complexity of MS is not easily replicated in rodent models of EAE which emphasises the distinct immunological, genetic, developmental and environmental differences between humans and laboratory rodents. This can be somewhat overcome with the use of humanised mouse models.
Other ways include the use of non-human primates, including the rhesus macaque and the common marmoset, to attempt to replicate the human disease.
The first marmoset EAE model was first described over 20 years ago. Induction with whole human myelin causes a 100% disease incidence and a chronic relapsing-remitting disease course alongside mononuclear infiltration, demyelination and reactive astrogliosis. Further development through the years has resulted in models with increased disease incidence and lower variability. Their outbred nature better reflects the heterogeneity seen in the MS population.
These marmoset models can be used in preclinical trials and are particularly useful for assessing the therapeutic potential and safety of candidate drugs. Despite these obvious advantages, the ethical and economical implications of using non-human primate models mean that they are rarely used in preclinical research.
For more information on the animal models of MS :
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7094661/https://www.frontiersin.org/articles/10.3389/fnmol.2022.1017484/full
https://onlinelibrary.wiley.com/doi/10.1111/bpa.12454
https://www.sciencedirect.com/science/article/abs/pii/B9780128094686000371
Understanding the brain & harnessing the body’s repair system
Repairing nerve cells
In order to treat the disease properly, it is important to understand it. Dr Anna Williams from the Centre of Regenerative Medicine in Manchester is particularly interested in trying to understand how the brains of patients with MS repair. Indeed, the brain has the possibility to put myelin sheaths back onto nerves, which has the potential to restore nerve function in MS. However, this process is generally inefficient, and often forms scars which can cause long-term problems for patients. Cells that have this repair function are called oligodendrocytes. The research team has looked into how these cells work and whether they could get them to repair myelin damage more effectively.
By growing oligodendrocytes in dishes, it is possible to monitor whether they make more or less myelin depending on different conditions or drugs. The team does a lot of experiments using cells from mice, rats and humans and slices of brain from mice and rats. “Ultimately, the work moved to live animals. As humans are the only animals that get MS naturally, we therefore tried to model part of the disease in mice. We’re interested in myelin repair, so we caused a small amount of demyelination in the animals by chemical-injection in the brain. The mice don’t seem to be affected by this. Mice repair their brains much better than humans do. So within 4 weeks, basically all signs that myelin damage goes away,” explains Dr Anna Williams. “The mice don’t develop symptoms but imaging of the brains can give us a before/after treatment picture to see how well oligodendrocytes are repairing myelin. We measure the pathology rather than the pain.”
There are currently three drugs that have entered human trials for trying to improve myelin repair in MS after studies in animals. Dr Anna Williams worked on one of them called Baroxatine.
Fibrin
Looking into the repair system of nerves, researchers have also identified a molecule of interest. Fibrin, a protein that helps form blood clots, may inhibit the repair of damaged nerves. Previous research had shown that fibrin accumulates in the damaged nerves of MS patients, but research in mice has recently shown that fibrin-free mice regenerate myelin faster than normal mice after damage to the sciatic nerve in the leg5. In 2004 researchers found that EAE mice that were bred without fibrin showed a later disease onset and lived longer that normal EAE mice6.
CNTF & TNF- α
Researchers also found that EAE mice had a more severe form of the disease if they lacked a chemical messenger known as CNTF. They proposed that CNTF protects oligodendrocytes against programmed cell death through a mechanism that involves a substance called tumour necrosis factor alpha (TNF-α)7 8. Other scientists found that administration of another messenger, LIF, can reverse this loss of oligodendrocytes normally seen in EAE. This compound is well-tolerated in humans9.
Paradoxically, MS does not respond to TNF-α, and it has now been shown that TNF-α has two opposite effects in mice, first accelerating damage to nerves and later accelerating its repair. This finding may help the development of more effective treatments for the disease. In MS, cells have some capacity for self-repair, so a promising treatment approach is to increase the body’s ability to repair itself10.
Stem cells and transplants
Lymphocyte transplants
An emerging potential treatment removes the ‘self-reactive’ lymphocytes, replacing them with lymphocytes developed from the patient’s own stem cells. In effect, this is a recalibration of the person’s immune system which removes the abnormal aspects and transplants their own immune cells back into the body. Because the replenishing white cells are developed from the patient’s own stem cells, they do not induce the usual problems of rejection.1
Glial cell transplants
A new study shows that when specific human brain cells are transplanted into animal models of multiple sclerosisand other white matter diseases, the cells repair damage and restore function. The findings are the culmination of more than 15 years of research understanding support cells found in the brain called glia, how the cells develop and function, and their role in neurological disorders.
When human glia progenitor cells are transplanted into adult mouse models of progressive multiple sclerosis, the cells migrated to where needed in the brain, created new oligodendrocytes, and replaced the lost myelin. The study also showed that this process of remyelination restored motor function in the mice. The findings, which appear in the journal Cell Reports, are the culmination of more than 15 years of research at URMC understanding support cells found in the brain called glia, how the cells develop and function, and their role in neurological disorders.
Bone marrow transplants
HSCT (haematopoietic stem cell transplantation) is an intense chemotherapy treatment for MS. It aims to 'reset' the immune system by wiping it out and then regrowing it, using your stem cells. The use of HSCT to treat autoimmune disorders was first considered following observations of autoimmune disease remission when bone marrow tissue was destroyed during cancer therapy.
Researchers found that bone marrow transplants could induce either resistance or susceptibility to autoimmune diseases, and realised that the bone marrow cells were key to the development of such diseases. However, the destruction of bone marrow was an aggressive treatment regime which severely depleted the patient’s immune system. Using animal models, researchers were able to conclude that bone marrow transplants were a viable treatment for MS, but that severe depletion of lymphocytes also raised the mortality risk.2 These results led to the development of less aggressive conditioning regimes which would be safer for human patients. Researchers are also using animal models to further investigate whether the source of the HSCT should be the individual concerned, or a donor.3
Targeting the immune system
In 2004 scientists reported that inhibiting a protein called Nogo A in EAE mice significantly delays the onset of the condition11 12 13. It may therefore play a role in the human disease.
Two immune proteins found in mouse brains raise the possibility that autoimmune diseases such as MS may be the result of a misguided attack by the immune system on the nerve cells that carry 'genetic fingerprints'14. Such attacks are carried out by subset of white blood cells called T cells.
A specially engineered CD28 molecule blocks interactions between immune-system and suppresses EAE disease in mice. It interferes with the interaction that prompts T-cells to attack nerve cells in MS15. Researchers have found a way of using normal T-cells to recognise misdirected T-cells in EAE mice. Mice treated in this way survived, whereas 50% of the untreated EAE mice died16.
In MS, T-cells in the immune system are stimulated to attack myelin sheaths when they are exposed to small amounts of myelin protein, while T-cells exposed to large amounts of the same protein will self-destruct. This has led to the development of immunological therapy, which has been tested successfully in marmosets17. T-cells are, however, essential to survival, so researchers have devised a way to eliminate only those T-cells that are causing the problem. A molecule that targets a specific type of T cell delivers a toxic drug called doxorubicin. This might prove useful in several autoimmune diseases18.
Using laboratory cell tissue cultures and mice genetically manipulated to lack two key immune system cells, researchers showed that Mycobacterium leprae, the leprosy bacterium, destroys the protective myelin sheath that surrounds nerve fibres and then hides in Schwann cells, poised to initiate later attacks. This novel bacterial mechanism of inducing demyelination may provide clues to early molecular events in MS19.
Three potential drug treatments
MS patients have toxic levels of glutamate in their spinal fluid. Glutamate can cause extensive brain damage following stroke. Two studies have shown that compounds that block glutamate, which were being tested as stroke treatments, also protected rats and mice with EAE25 26. Similar compounds may prove useful in the treatment of MS.
Two anti-diabetic drugs called TZDs, already approved in the USA for treatment of Type 2 diabetes, prevented MS in mice and treated those already ill. There have already been small scale successful trials of one of these, pioglitazone, in humans. Even if the drugs are only as good as those currently in use, they still offer an advantage because they can be taken orally27.
Many MS patients are adamant that cannabis relieves their tremors and muscle spasms, and research in mice confirms this. When EAE mice were injected with cannabis extracts, these symptoms improved within minutes and did not return for hours. The researchers found that the cannabis stimulated certain receptors on the surface of motor nerve cells and did not have a sedative effect28.
A team managed to reduce chronic inflammation associated with multiple sclerosis in mice thanks to the administration of a type of lipid that mediates inflammation. The team found that these types of mediator substances, responsible for resolving the inflammatory process when it is no longer beneficial, are minimized in people with multiple sclerosis as well as in animal models of the disease. The use of these mediators could become a good strategy for the treatment of this autoimmune disease.
Diagnosis - Finding biomarkers of the disease
MS is a complex disease characterised by important pathophysiological heterogeneity affecting the clinical appearance, progression and therapeutic response for each patient. To date, there is no specific molecular test for the treatment choice, which is based more on risk assessment than on the specific needs of the patients. Therefore, there is a strong unmet need to define specific biomarkers that will reflect the different features of the disease. This will facilitate the prediction of the clinical outcome and the therapeutic choice among the growing number of treatments available for MS.
There are five groups of clinically relevant MS molecular biomarkers, based on the key pathological hallmarks of MS: inflammation, blood–brain barrier disruption, myelin and axonal damage, gliosis and, ultimately, repair mechanisms. In many cases, early-stage biomarkers are first discovered and studied in animal models and then validated in human disease to evaluate their potential for diagnosing, predicting or treating human disease.
Like any disease animal model, EAE models can sometimes fail to mimic the entire spectrum of human disease, but they can nonetheless recapitulate the disease’s primary hallmarks. In fact, the EAE model is a valuable tool for understanding MS physiopathological mechanisms and for identifying biomarkers fundamental for drug development. The different biomarkers recapitulating the main hallmarks of the disease are comparably dysregulated in EAE models and MS. Biomarkers expression between EAE models and MS disease often coincides, making the EAE model a valuable tool for biomarker discovery and it will most likely continue to provide MS drugs.
https://hal.science/hal-04078010v1/preview/islandora_162072.pdf
Diagnosis
https://www.sciencedaily.com/releases/2022/03/220324122612.htm
Origin of the disease
Comparing animal and human study results has revealed that human autoimmune diseases are caused by multiple underlying factors. Animal models have shown genetic factors to be strongly influential, whereas in humans, it is suggested that environmental factors may be key to the development of the disease.
Genetic aspects
Mice play a key role in allowing understanding of the genetic component of MS20. A gene in mice, similar to a corresponding human gene, has been shown to be important in making myelin, and the events that lead the immune system to destroy myelin are being worked out by studying mice21. Understanding how myelin is made and destroyed and how it can be replaced when lost may lead to effective therapies.
A critical gene, osteopontin, is a key factor in the inflammatory immune response and may determine the cycle of relapses and remissions. In rodents, disease severity is proportional to the amount of osteopontin gene expression. Osteopontin-deficient mice have milder disease and more frequent remission. The findings could lead to targeted new therapies for MS. However, more research is needed, especially as other genes are involved in the disease22. Indeed, when researchers analysed brain tissue from four MS patients, they found that the expression of 39 genes was increased, and the expression of 49 other genes was decreased. They selected two of these genes for therapeutic testing in mice with EAE, and found that the disease was less severe in mice deficient in the antibody receptor Fc, and that a naturally-occurring substance called G-CSF improved the condition23.
Two genes, known as Olig1 and Olig2, control the development of cells that the central nervous system uses to generate muscle movement. Mice bred to lack one or both Olig genes failed to develop the motor nerves needed to control muscle movement and were completely paralysed. Olig genes induce oligodendrocytes to surround and protect the nerves with myelin24.
Recent discoveries regarding MS : https://www.sciencedaily.com/news/health_medicine/multiple_sclerosis/
https://www.mssociety.org.uk/research/latest-research/latest-research-news-and-blogs
References
References from the earlier version of this page. Modern references provided as links and within the body of the article.
- Burt RK, Loh Y, Cohen B, et al. Autologous non-myeloablative haemopoietic stem cell transplantation in relapsing-remitting multiple sclerosis: a phase I/II study. Lancet Neurol 2009; published online January 30.
- Burt RK, Traynor AE. Hematopoietic stem cell transplantation: a new therapy for autoimmune disease. Oncologist. 1999;4(1):77-83
- Sykes M, Nikolic B. Treatment of severe autoimmune disease by stem-cell transplantation. Nature. 2005 Jun 2;435(7042):620-7. Review.
- MS Society, 2009. Stem cell therapy may halt and even reverse disability in people with relapsing remitting MS. (Research News) [Online] (Updated 30 Jan 09). Available at: http://www.mssociety.org.uk/research/news_in_research/research_news/stem_cell_trial.html
- Akassoglou K, Yu W-M, Akpinar P, Strickland S (2002) Fibrin inhibits peripheral nerve remyelination by regulating Schwann cell differentiation Neuron 33, 861
- Akassoglou K, Adams RA, Bauer J et al (2004) Fibrin depletion decreases inflammation and delays the onset of demyelination in a tumor necrosis factor transgenic mouse model for multiple sclerosis. Proc Nat Acad Sci 101, 6698
- Akassoglou K, Adams RA, Bauer J et al (2004) Fibrin depletion decreases inflammation and delays the onset of demyelination in a tumor necrosis factor transgenic mouse model for multiple sclerosis. Proc Nat Acad Sci 101, 6698
- Linker RA, Mäurer M, Gaupp S et al (2002) CNTF is a major protective factor in demyelinating CNS disease: A neurotrophic cytokine as modulator in neuroinflammation Nature Medicine 8, 620
- Butzkueven H, Zhang J-G, Soilu-Hanninen M et al (2002) LIF receptor signaling limits immune-mediated demyelination by enhancing oligodendrocyte survival Nature Medicine 8, 613
- Arnett HA, Mason J, Marino M et al (2001) TNF promotes proliferation of oligodendrocyte progenitors and remyelination Nature Neuroscience 4, 1116
- Jameson BA, McDonnell JM, Marini JC & Korngold R (1994) A rationally designed CD4 analogue inhibits experimental allergic encephalomyelitis Nature 368, 744
- Lonberg N, Taylor LD, Harding FA, Trounstine M et al (1994) Antigen-specific human antibodies from mice comprising four distinct genetic modifications Nature 368, 856
- Karnezis T, Mandemakers W, McQualter JL et al (2004). The neurite outgrowth inhibitor Nogo A is involved in autoimmune-mediated demyelination. Nature Neuroscience 7, 736
- Huh GS, Lisa M. Boulanger, Du D et al (2000) Functional Requirement for Class I MHC in CNS Development and Plasticity Science 290, 2155
- Srinivasan M, Gienapp IE, Stuckman SS et al (2002) Suppression of Experimental Autoimmune Encephalomyelitis Using Peptide Mimics of CD28 J Immunol 169, 2180
- Jyothi MD, Flavell RA, Geiger TL (2002) Targeting autoantigen-specific T cells and suppression of autoimmune encephalomyelitis with receptor-modified T lymphocytes Nature Biotech 2, 1215
- McFarland HI, Lobito AA, Johnson MM et al (2001) Effective antigen-specific immunotherapy in the marmoset model of multiple sclerosis J Immunol 166, 2116
- Casares S, Stan AC, Bona CA, Brumeanu T-D (2001) Antigen-specific downregulation of T cells by doxorubicin delivered through a recombinant MHC II-peptide chimera Nature Biotechnology 19, 142
- Rambukkana A, Zanazzi G, Tapinos N, Salzer JL (2002) Contact-dependent demyelination by Mycobacterium leprae in the absence of immune cells Science 296, 927
- Ebersole TA, Chen Q, Justice MJ, Artzt K (1996) The quaking gene product necessary in embryogenesis and myelination combines features of RNA binding and signal transduction proteins Nature Genetics 12 (3), 260
- Klein L, Klugmann M, Nave K et al (2000) Shaping of the autoreactive T-cell repertoire by a splice variant of self protein expressed in thymic epithelial cells Nature Med 6, 56
- Chabas D, Baranzini SE, Mitchell D et al (2002) The influence of the proinflammatory cytokine, osteopontin, on autoimmune demyelinating disease Science 2001 November 23; 294, 1731
- Lock C, Hermans G, Pedotti R et al (2002) Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis Nature Medicine 8, 500
- Lu R, Tao Sun, Zhu Z, Nan Ma N et al (2002) Common developmental requirement for olig function indicates a motor neuron/oligodendrocyte connection Cell 109, 75
- Smith T, Groom A, Zhu B et al (2000) Autoimmune encephalomyelitis ameliorated by AMPA antagonists Nature Med 6, 62
- Pitt D, Werner P, and Raine C (2000) Glutamate excitotoxicity in a model of multiple sclerosis Nature Med 6, 67
- Feinstein D, Galea E, Gavrilyuk V et al (2002) Perixisome proliferator-activated receptor- agonists prevent experimental autoimmune encephalomyelitis. Ann Neurol 51, 694
- Baker D, Pryce G, Croxford JL et al (2000) Cannabinoids control spasticity and tremor in a multiple sclerosis model Nature 404, 84
Last edited: 31 May 2023 09:48
